Research Projects
Latency Reversal
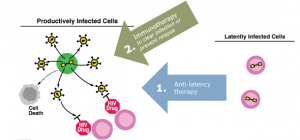
Curing HIV infection has been a difficult challenge primarily due to the persistence of viral reservoirs that remain in the body even during uninterrupted, long-term antiretroviral therapy. These reservoirs exist as HIV genomes (proviruses) that integrate into the cellular DNA of extremely long-lived immune cells known as T cells. Within these cells, the provirus can remain silent and undetectable for decades in a state known as viral latency. During this time, the provirus enjoys sanctuary from drug therapy and from the host immune system. If therapy is interrupted at any point, the provirus can reactivate, resulting in viral rebound within the body. Without intervention, increased viral loads can lead to depletion of host T cells, impairment of the immune system, progression to AIDS, and ultimately death. Targeted approaches are being explored to reverse latency, induce viral antigen expression within formerly latently infected cells, and to pair the latency reversing agents with immune clearance mechanisms to eradicate persistent infection. One of our research goals is to understand how to reverse latency so that the virus can be cleared from the body.
UNC HIV Cure Center has a rich and diverse latency reversal research portfolio with two major themes. The first is the discovery, development and testing of latency reversing agents. Libraries of compounds are being screened to assess their ability as single agents or in combination to reverse latency. Hits from these screens are moved forward into in vitro cell line and primary cell models and if warranted, in vivo models The second is the identification of new targets that can be exploited to selectively re-activate HIV. Cellular pathways that enforce HIV latency are good targets for agents that reverse latency and we are exploring these pathways to identify druggable targets.
Clearance
Ferrari, Nature Reviews Drug Discovery, 2016
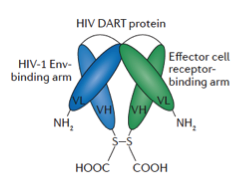
Targeted approaches to reverse latency are being explored so that viral antigen is expressed by a formerly latently infected cell and becomes vulnerable to immune clearance mechanisms. We are exploring antibodies, engineered antibody-like Dual Affinity Re-Targeting (DARTs) molecules, vaccines, and immune checkpoint modulation strategies to clear the HIV virus from the body and the UNC HIV Cure Center has a rich and diverse clearance research portfolio centered around these major approaches.
Therapeutic antibodies that can specifically direct immune clearance of HIV-infected cells are a relatively new area of HIV cure research and may be useful and important tools for clearance. DARTs are designed to redirect the activity of T-cells to target the cells infected with HIV. Novel immune-augmenting strategies, such as adjunctive treatment with immune checkpoint inhibitors, might reawaken an extant but exhausted or diminished immune response and facilitate clearance of viral reservoirs. T-cell vaccines are also being tested for their ability to redirect the immune response to enhance clearance. These clearance strategies are being tested alone and in combination with latency reversing agents in in vitro and in vivo models.

